One critical reality defines today’s US MedTech market: the winners are not those who promise the most innovation, but those who use it reliably.
As care moves toward telemedicine, remote diagnostics, and personalized home-based solutions, software becomes the foundation of device performance. This tendency creates an opportunity for medical device manufacturers, but it also increases technical and regulatory complexity.
Understanding how software architecture choices shape compliance, security, and long-term product performance is now a strategic priority for MedTech leaders. Below, we explore the key forces redefining software design in the US market and their implications for your next product cycle.
Key trends shaping the US MedTech landscape
Site-of-care shift
Rising healthcare costs in the US, 17.6% of GDP, reshape how and where care is delivered. Now, both payers and consumers are prioritizing prevention, remote monitoring, and earlier diagnostics, with homes becoming a primary site of care.
A critical but often overlooked driver of this shift is the insurance industry itself. A preventive diagnosis is much cheaper and faster than a reactive one. If a patient relapses or complications go undetected, insurers pay again, and that directly affects their margins.
Thus, the US remote patient monitoring (RPM) market continues to expand. Tools like cardiac monitoring patches, glucose sensors, cough sound recognition tools, and connected blood pressure cuffs are moving from pilot programs into scalable deployment.
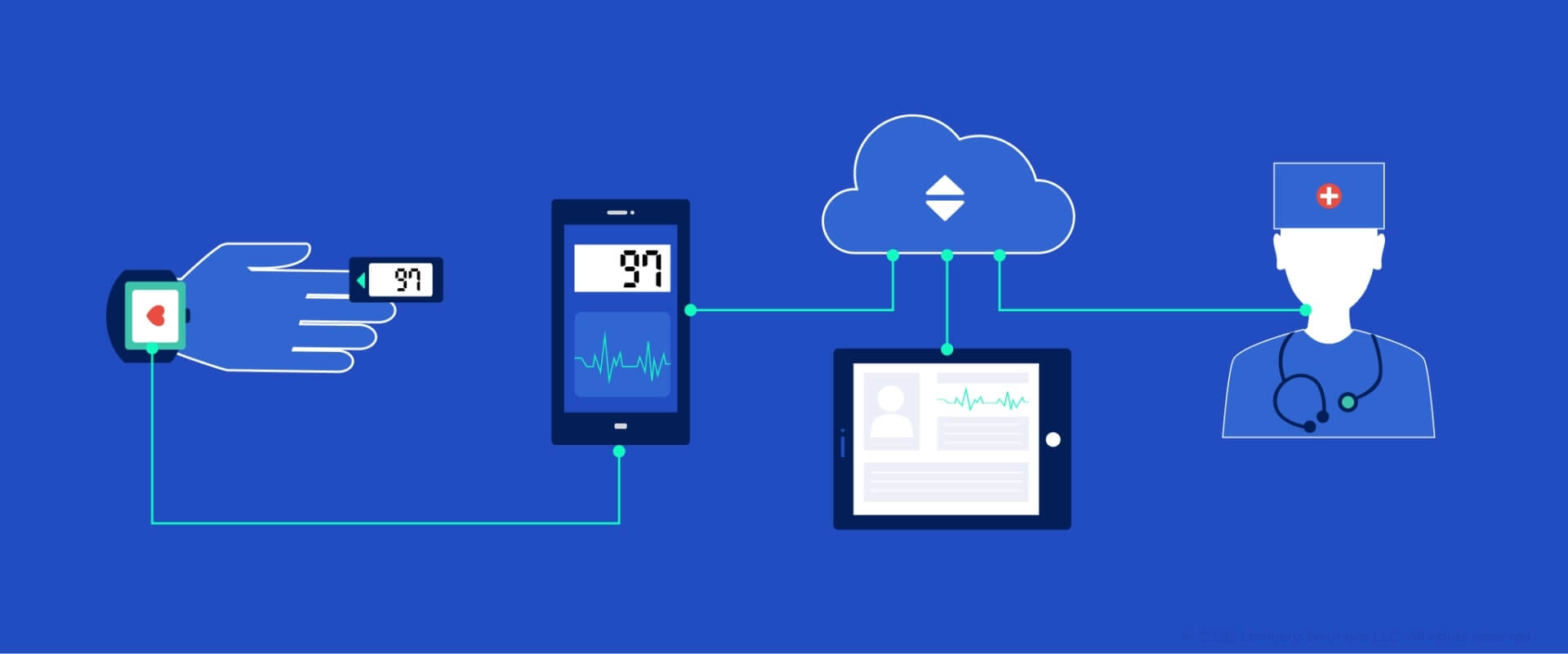
For manufacturers, this shift creates clear business requirements: devices must be energy-efficient, secure, interoperable with EHR and EMR systems, and reliable in non-clinical environments. And the opportunity lies not just in building devices, but in architecting compliant, connected ecosystems that function seamlessly in patients’ homes.
Consumerization of care
Every second adult in the US uses at least one digital health tool, from fitness trackers to remote monitoring apps. So, patients expect mobile-first devices with real-time feedback, clean interfaces, and actionable insights without complex manuals or clinic-dependent workflows. For clinicians, these devices should also be interoperable with EHR systems and ensure reliable performance in high-stakes environments.

This creates a dual mandate. Devices must deliver a consumer-grade user experience while maintaining clinical-grade reliability, cybersecurity resilience, and full compliance with FDA quality system requirements. That requires sophisticated expertise in all stages of medical software development, from design to secure cloud architecture and data governance.
AI and data integration
The US MedTech market also embraces AI adoption, not only for diagnostics but also for workflow automation and operational optimization. The examples are remarkable: AI reading scans to spot cancer signs, AI-powered symptom checkers, and skin disease monitoring tools. However, the AI implementation level still remains conservative compared to other regulated industries. While interest in AI is high, full-scale deployment remains limited due to concerns about data security, validation, bias, and regulatory uncertainty.
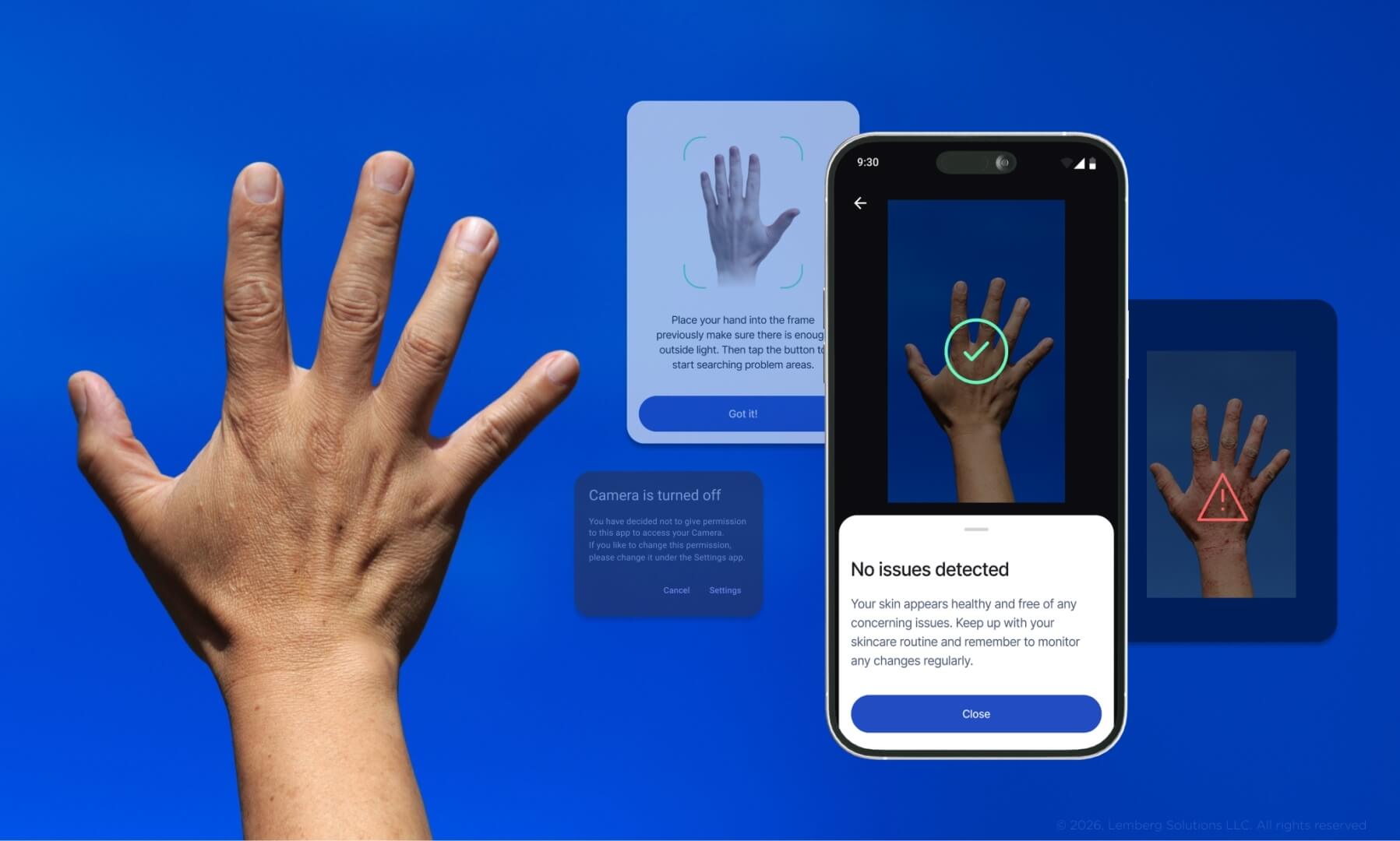
From a business perspective, this creates a gap between marketing demand and the reality of implementation. Many companies position themselves as “AI-powered,” yet few have moved beyond pilot programs or limited clinical applications.
Scaling AI in MedTech requires far more than model accuracy. It demands high-quality data (which is often confidential), FDA and HIPAA compliance, strong cybersecurity controls, integration with legacy hospital IT systems, and clear clinical accountability. Without these foundations, AI remains a feature in a slide deck rather than a commercially viable solution.
The real competitive advantage will belong not to those who market AI the loudest, but to those who industrialize it. They embed governance, validation, security, and lifecycle management into their product architecture from day one.
Robotics and automation
Automation is no longer limited to surgical robotics. It now extends across the entire medical device lifecycle, from design and manufacturing validation to intraoperative performance. For example, industrial CT systems are increasingly used during production to detect internal defects, verify structural integrity, and generate objective evidence for regulatory submissions.
This shift reflects a broader priority: reducing recall risk, strengthening compliance documentation, and preventing costly post-market failures.

This dual expansion of robotics and automation fundamentally reshapes software design priorities. That raises the bar for system reliability and secure lifecycle management. And works in both patient-facing applications and in manufacturing environments that ensure device safety before deployment.
Microextrusion system and 3D printing
US MedTech also embraces microextrusion and 3D printing as key enablers for the manufacturing of medical components: catheters, drug-delivery tubing, and minimally invasive device shafts

The FDA recognizes additive manufacturing as a pathway for producing custom, patient-matched devices and complex geometries that traditional manufacturing cannot easily achieve. As demand for personalization grows, however, so does operational complexity. Sіcaling microextrusion and 3D printing requires disciplined design controls, validated materials, documented process parameters, and full traceability.
Key challenges US MedTech companies face
Along with opportunities, the digitalization of healthcare brings challenges. High regulatory scrutiny, litigation exposure, cybersecurity risks, and reimbursement pressure create an environment where engineering missteps quickly become financial setbacks. For manufacturers, growth opportunities are real, yet so are the structural risks that can stall or derail commercialization.
Cost of FDA delays
First, the cost of FDA delays is always high. Regulatory approval timelines directly affect revenue and market positioning. When FDA submissions lack complete verification evidence, cybersecurity documentation, traceability matrices, or risk management files, the software development lifecycle extends. These delays can increase burn rate and postpone product launches, allowing competitors to gain a first-mover advantage.
The risk of software failures
Second, the risk of recalls due to software failures is always high, too. As medical devices become software-centric, the probability of firmware defects, cybersecurity vulnerabilities, or integration failures increases. A single software flaw can trigger FDA safety communications, mandatory corrective actions, and even full recalls. Beyond direct remediation costs, recalls damage brand credibility and may expose companies to liability claims.
Lack of expertise
Third, developing FDA-compliant, secure, and scalable medical device software requires cross-functional expertise in the firmware engineering lifecycle. These specialized skills are in short supply and expensive in the US market. As a result, many MedTech companies struggle to scale innovation while maintaining compliance discipline.
Taken together, these challenges reveal a broader reality: technical execution is inseparable from business performance. And regulatory readiness, software reliability, and cybersecurity maturity are strategic levers that influence valuation, speed to market, and long-term trust.

Strategic implications for MedTech leaders: designing for compliance, scale, and trust
Having a software architecture strategy in place from day one supports secure-by-design principles and ensures scalability as the product evolves. The right architecture decisions define how you track requirements, manage updates, protect data, control cybersecurity risks, and connect with other systems. When these elements are added late in development, teams often face rework, delays, and higher compliance costs.
However, the US recognizes that the challenge is not only in maintaining control but also in retaining qualified engineers. In-house medical software development often results in prolonged hiring cycles, overloaded internal teams, and extended development timelines. In this case, a carefully selected healthcare software development company can alleviate these issues while allowing manufacturers to retain product ownership and regulatory accountability.
Security and compliance are highly important in this context, as they influence purchasing decisions. Hospital procurement teams assess cybersecurity posture. Investors evaluate regulatory maturity. Only companies that treat security, validation discipline, and documentation rigor as architectural principles succeed.
The bottom line
The US MedTech market is evolving into a software-defined industry, where device performance and reliability determine competitive position. At the same time, regulatory expectations are becoming more demanding, increasing the operational burden on development teams.
In this environment, strategic contract engineering partnerships allow manufacturers to extend their internal capabilities without overextending their organizational structure. In a market where experienced medical software developers are in short supply, external engineering teams provide immediate access to domain-specific expertise that would otherwise take months to recruit and onboard.